Studies
Look at Liver Fibrosis in HIV-HCV and HIV-HBV Coinfected People By
Liz Highleyman
Over
years or decades, chronic hepatitis B virus (HBV)
or hepatitis C virus (HCV) infection can progress
to advanced liver disease including cirrhosis
and liver cancer.
Some evidence indicates that this process may happen faster in HIV positive people
-- especially those with low CD4 cell counts -- but data have been inconsistent.
Several
studies presented at the recent 16th Conference on Retroviruses
and Opportunistic Infections (CROI 2009) last month in Montreal shed further
light on fibrosis in HIV-HBV and HIV-HCV coinfected individuals. 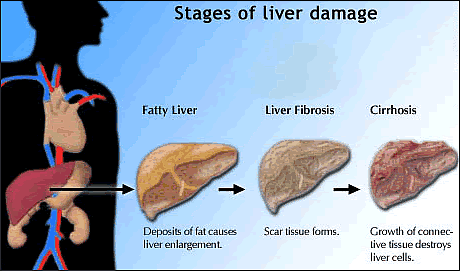
More
Fibrosis in HIV-HCV Coinfected Hemophiliacs
Margaret Ragni from
the University of Pittsburgh and colleagues compared liver disease progression
in 92 HIV-HCV coinfected and 128 HCV monoinfected hemophiliac men at 34 U.S. treatment
centers (participants in the HHH Hemophilia Cohort Study) who underwent liver
biopsies.
Among the 220 men with available biopsy findings, 86 (39%) had
some degree of fibrosis (Ishak scores > 3 or Metavir scores >
2). However, HIV-HCV coinfected participants were significantly more likely to
have fibrosis (47%) than HCV monoinfected individuals (32%; P = 0.03), despite
being about the same age (43 vs 40 years), being infected with HCV for a similar
duration (38 vs 36 years), drinking similar amounts of alcohol, and being about
equally likely to receive hepatitis C treatment (48% vs 41%). After controlling
for HIV status, older age --but not race, alcohol consumption, or lack of hepatitis
C treatment -- predicted worse fibrosis.
Average Ishak fibrosis scores
were 2.3 in the coinfected group versus 1.9 in the HCV monoinfected group (Metavir
1.6 vs 1.3), a difference that was marginally significant (P = 0.06 for Ishak,
0.045 for Metavir). HIV-HCV coinfected participants also had higher average AST
levels and lower average platelet counts than HCV monoinfected men.
The
investigators also measured levels of various cytokines to test the hypothesis
that HIV might accelerate liver disease progression through up-regulation of cytokines
that promote fibrosis. Coinfected men were more likely than HCV monoinfected men
to have high TFG-beta-2 levels, but less likely to have elevated interferon-gamma
(IFN-gamma). Levels of interleukin-6, interleukin-10, and cytokine promoter genotypes
did not differ according to HIV serostatus.
"Nearly half of HIV-HCV
coinfected hemophiliac men have Ishak > 3 fibrosis, a 1.5-fold higher
prevalence than in HCV monoinfected men," the investigators concluded. "Cytokine
dysregulation may contribute to fibrosis progression among those with HIV-HCV
coinfection, but other mechanisms likely affect fibrosis score." No
Fibrosis Difference in Texas Study
In another analysis of the
relationship between fibrosis and HIV infection, Mamta Jain and colleagues from
the University of Texas Southwestern Medical Center in Dallas performed a retrospective
cross-sectional analysis of liver biopsies from 180 HIV-HCV coinfected and 407
HCV monoinfected patients between 1998 and 2008. The HIV positive participants
had well-preserved immune function, with a median CD4 count of 459 cells/mm3 and
70% with undetectable HIV viral load. This
study classified biopsy samples using the Batts-Ludwig scale (mild fibrosis =
stage 0, 1, or 0-1; moderate = 1-2, 2, or 2-3; severe = 3-4 or 4). Here, HIV-HCV
coinfected and HCV monoinfected participants had a similar distribution of mild
(39% vs 34%), moderate (41% vs 49%), and severe fibrosis (18% vs 16%). Liver steatosis,
or fat accumulation, was slightly less common among HIV negative individuals (genotype
distribution did not differ, with about 90% having genotype 1). "The
distribution of fibrosis stage was similar in those with HCV compared to those
with HIV-HCV, which may be due to high CD4 cell counts and viral suppression of
HIV in the coinfected population," the researchers concluded; however, they
added that the coinfected patients were younger (median 43 vs 47 years) and "faster
progression of disease remains a possibility." In
a multivariate analysis adjusting for potential confounding factors, older age,
steatosis, and elevated AST were independent risk factors for moderate-to-severe
versus mild fibrosis, but HIV infection itself was not, leading the investigators
to suggest that "perhaps other factors besides HIV-infection may play a significant
role in the development of fibrosis." Effect
of HIV-HBV versus HIV-HCV Coinfection Paula
Tuma and colleagues from Hospital Carlos III in Madrid, Spain, looked at liver
disease outcomes in HIV positive individuals coinfected with HBV or HCV. This
longitudinal retrospective study included all 27 HIV-HBV coinfected patients seen
at their center who had undetectable HBV DNA while being treated with HAART that
had anti-HBV activity. [The nucleotide/nucleoside analog drugs lamivudine
(3TC; Epivir), emtricitabine
(Emtriva), and tenofovir (Viread,
also in the Truvada and Atripla
combination pills) have dual activity against both HIV
and HBV.] In addition, 46 patients with HIV
only and 148 HIV-HCV coinfected patients -- also treated with HAART
-- were used as comparison groups.
The median age was 47 years in the HIV-HBV
group and 44 years in the other 2 groups. Overall, patients had well-controlled
HIV with a median CD4 count of 528 cells/mm3 and HIV RNA < 50 copies/mL. At
baseline, median ALT levels tended to be higher in HIV-HCV coinfected compared
with HIV-HBV coinfected or HIV monoinfected patients, but the difference did not
reach statistical significance.
 The
investigators used transient elastometry (FibroScan), a method that estimates
fibrosis based on liver "stiffness." Changes in estimated fibrosis were
compared between the first and last examinations, done 2.0 to 3.6 years apart.
Baseline liver stiffness was 6.9, 5.6, and 6.8 kiloPascals (kPa) in the HIV-HBV,
HIV monoinfected, and HIV-HCV group, respectively, but again this was not a significant
difference. The
investigators used transient elastometry (FibroScan), a method that estimates
fibrosis based on liver "stiffness." Changes in estimated fibrosis were
compared between the first and last examinations, done 2.0 to 3.6 years apart.
Baseline liver stiffness was 6.9, 5.6, and 6.8 kiloPascals (kPa) in the HIV-HBV,
HIV monoinfected, and HIV-HCV group, respectively, but again this was not a significant
difference.
After a median 32 months of follow-up, HIV-HCV coinfected patients
had significantly higher median ALT (55 IU/L) than HIV monoinfected (34 IU/L;
P = 0.01) or HIV-HBV coinfected (30 IU/L; P = 0.02) participants. Median liver
stiffness was similar in the HIV-HCV and HIV-HBV coinfected groups (7.7 vs 7.4
kPa), but significantly lower in the HIV monoinfected group (5.4 kPa; P = 0.02).
About
twice as many HIV-HCV coinfected patients experienced liver disease progression
of at least 1 Metavir stage compared with HIV-HBV coinfected patients, who in
turn were about twice as likely to progress as the HIV monoinfected participants
(26.4%, 14.8%, and 6.5%, respectively). But median liver stiffness increased by
0.6 and 0.05 kPa in the HIV-HCV and HIV monoinfected groups, while slightly decreasing
by 0.2 kPa in the HIV-HBV coinfected group (P = 0.03).
In a multivariate
analysis adjusting for age, sex, baseline ALT, and baseline liver stiffness, HIV-HCV
coinfected patients showed significantly more fibrosis progression than those
with HIV-HBV coinfection (P < 0.05), with an even greater disparity compared
with the HIV monoinfected group. However, the difference between the HIV-HBV coinfected
and HIV monoinfected groups did not reach statistical significance (P = 0.3).
Based
on these findings, the researchers concluded, "Liver fibrosis progression
in HIV patients is only associated with HCV coinfection in the HAART era. The
use of anti-HBV active HAART regimens has halted and/or even reversed liver fibrosis
in HIV-HBV coinfected patients." However, given the greater likelihood of
progression in the HIV-HBV patients and the small numbers in these 2 groups, a
deleterious effect of HIV-HBV coinfection cannot be ruled out. Spanish
GRAFIHCO Study
Juan Pineda from Hospital Universitario de Valme
in Seville, Spain, and colleagues also used the transient elastometry in a cross-sectional
analysis of liver fibrosis among 1310 HIV-HCV coinfected patients at 95 institutions
throughout Spain participating in the GRAFIHCO study (individuals who were triply
infected with HBV were excluded). Using this non-invasive method, the researchers
noted, allowed them to include more patients than typical biopsy-based studies.
Out
of the 1310 patients, 524 (40%) had a liver stiffness > 9 kPa, indicating
"significant" fibrosis, while 255 (19%) had liver stiffness >
14 kPa, indicating cirrhosis. Factors independently associated with significant
fibrosis were older age (adjusted odds ratio [OR] 1.04; P 0.002), heavy alcohol
consumption (> 50 g/day during the past 5 years)(OR 1.58; P = 0.013), and duration
of HCV infection (OR 1.03; P = 0.023).
Having a CD4 count below 200 cell/mm3
was also marginally associated with greater fibrosis (OR 1.67; P = 0.053), which
the researchers said supports earlier initiation of antiretroviral
therapy in this setting. On the other hand, having HCV genotype 4 was marginally
associated with less fibrosis (OR 0.66; P = 0.066), a finding they said "will
deserve further investigation." Benefits
of Anti-HCV Treatment
Finally, Jose Del Valle and colleagues
-- members of the same research collaboration as the study above -- looked at
the effect of hepatitis C treatment on liver fibrosis as estimated by transient
elastometry. Here, the investigators said that the non-invasive method might allow
for easier monitoring of treatment outcomes compared with repeated sequential
biopsies.
This prospective analysis included 50 HIV-HCV coinfected and
30 HCV monoinfected patients who started treatment
with pegylated interferon alfa-2a
(Pegasys) or alfa-2b (PegIntron) plus ribavirin at 2 Spanish hospitals; overall,
59% had hard-to-treat HCV genotypes
1 or 4.
All participants underwent transient elastometry at baseline
and 24 weeks after the scheduled end of treatment (the point at which sustained
virological response, or SVR, is determined). After completion of treatment, liver
stiffness decreased from a median 7.6 kPa at baseline to 6.6 kPa (P = 0.001),
with a median decrease of 0.50 kPa.
In a multivariate model that included
genotype, type of pegylated interferon, and length of anti-HCV therapy, only higher
baseline liver stiffness (P < 0.001) and achievement of SVR (P = 0.005) were
significant predictors of decreased liver stiffness after treatment.
"Liver
stiffness decreases significantly in HCV-infected patients treated with pegylated
interferon and ribavirin, especially in those with higher liver stiffness prior
to treatment and those who achieved sustained virological response," the
investigators concluded. "Transient elastometry can be a useful tool for
monitoring changes in liver fibrosis in HCV-infected patients receiving antiviral
therapy." 3/13/09 References M
Ragni, M Nalesnik, L Qin, and others. Cytokines and Liver Fibrosis in HIV/HCV
Co-infection. 16th Conference on Retroviruses and Opportunistic Infections (CROI
2009). Montreal, Canada. February 8-11, 2009. Abstract 830. M
Jain, E Neak, M Limerick, and others. Prevalence of Fibrosis Similar in Those
with HIV/HCV Compared to HCV Alone. CROI 2009. Abstract 831. P
Tuma, L Martin-Carbonero, P Barreiro, and others. Liver Fibrosis Progression in
HIV/HBV-co-infected Patients in the HAART Era. CROI 2009. Abstract 832. J
Pineda, C Tural, E Ortega, and others. Prevalence and Factors Associated with
Significant Liver Fibrosis Measured by Transient Elastometry in HIV/HCV-co-infected
Patients. CROI 2009. Abstract 824. J
Del Valle, A Camacho, J Mira, and others. Evolution of Liver Stiffness Evaluated
by Transient Elastometry in Patients Treated with Pegylated Interferon + Ribavirin.
CROI 2009. Abstract 826. |
|